|
Product Category : |
OXALATES |
|
Product :
BARIUM OXALATE
|
Product Code :
L015 |
Barium Oxalate is a derivative of Oxalic Acid. Barium Oxalate is practically insoluble in water and converts to the oxide when heated (calcined). Barium Oxalate is a White odourless powder. Barium Oxalate decomposes at the temperatures above 400oC. It should be stored in tightly closed containers in a Room Temperature.
Synthesis / Manufacturing Process of Oxalates
The major raw material to all the Oxalates is Oxalic Acid. Oxalic Acid can be prepared by oxidation of Carbohydrates or Glucose in presence of Nitric Acid & using Vanadium Pentoxide as a Catalyst. It can be also prepared by heating sodium formate in the presence of an alkali catalyst to form Barium Oxalate, which can be converted to free oxalic acid when treated with Sulphuric acid. However today Oxalic Acid is commercially prepared from Molasses route from cane molasses or sugarcane syrup.
Barium Oxalate is produced by reaction between Barium Hydroxide & Oxalic Acid. It can also be prepared by using Oxalic Acid & Barium Chloride as reactants.
Specifications :
|
CAS No. |
516-02-9. |
|
HSN No. |
29171100. |
|
Synonyms |
Barium Salt of Oxalic Acid. |
|
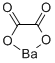
Molecular Formula |
C2BaO4
|
|
Molecular Structure |
|
|
Molecular Weight |
225.35. |
|
Appearance |
White odourless Powder. |
| Solubility | Practically insoluble in water. |
| Assay | 98.0% Min. |
| Packing | 25 Kg. HDPE Bags / Drums. |
Uses / Application of Barium Oxalate
Barium Oxalate is used as a green pyrotechnic colorant in specialized pyrotechnic compositions contain Magnesium.
|
To Know more about us, Visit our website :
www.newalliance.co.in
|